Soundbite Medical’s Directional Intravascular Lithotripsy Solution

The use of shock waves to treat calcium deposits, also known as lithotripsy, is a proven, powerful tool used by clinicians in patients across multiple clinical applications. Soundbite Medical has developed a unique platform for generating and delivering shock waves to vascular calcific lesions.
Soundbite’s Unique Mechanism of Action
Multiple, small amplitude mechanical shock waves are created outside of the body in the console
By adjusting the timing of the shock waves, they combine into one single, larger amplitude shock wave
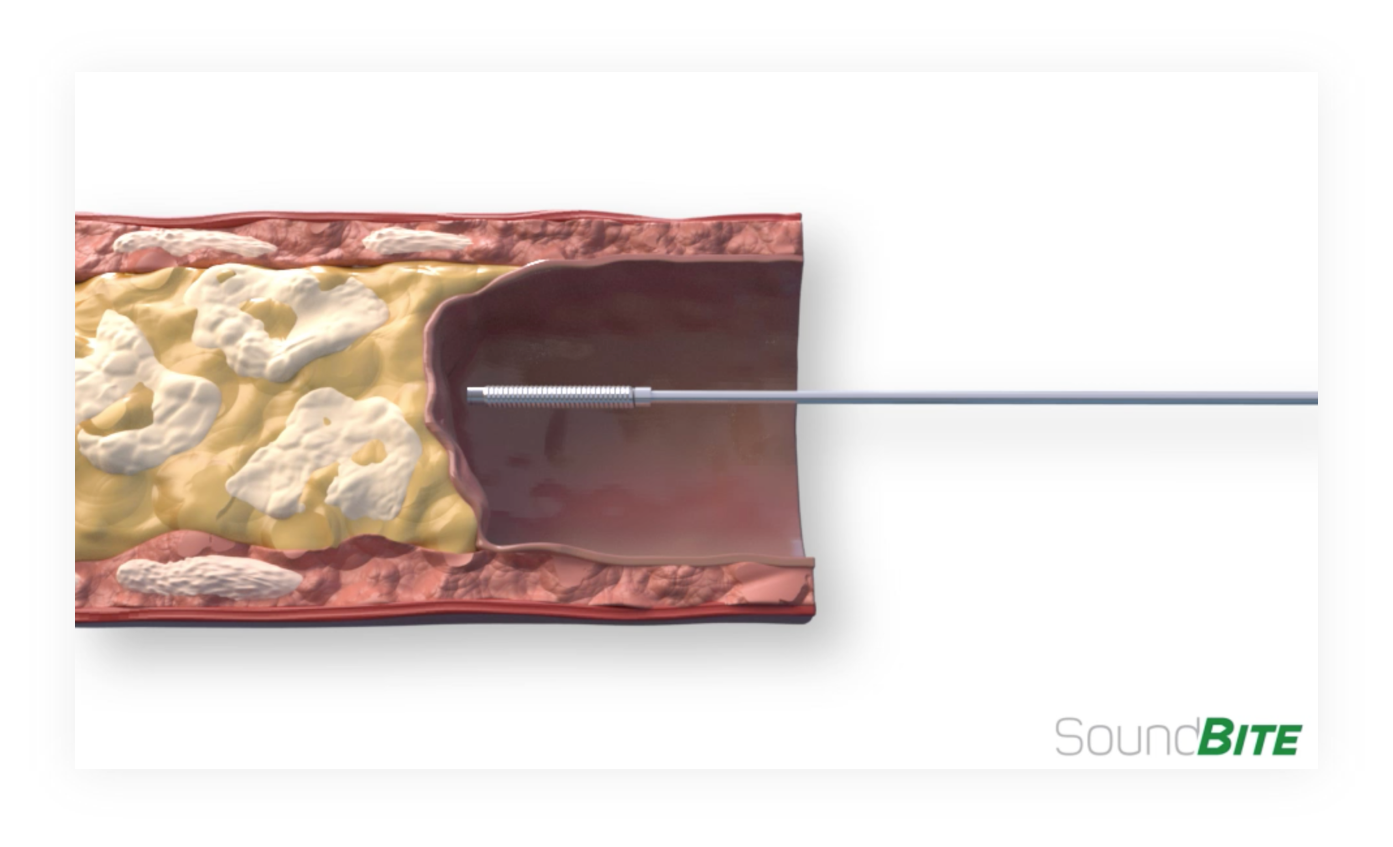
The single shock wave is delivered via an “Active” Waveguide Platform to the lesion inside the artery
As the shock wave leaves the waveguide, the resulting tip motion creates a micro jackhammer effect with the wire and the shock wave propagates and selectively penetrates and cracks the hard, calcific plaque while passing through the soft tissue

An example of how the shock wave propagates along the waveguide

Shock wave energy creates a hole in the “hard shell” of the egg…
…but is safe on soft tissue

SoundBite Active Wire unable to cross until “activated”
SoundBite XS Crossing System
It is designed to deliver shock waves to create a channel through the calcific cap or other calcified refractory barriers within a CTO. The Active Wire is inserted into the arterial vasculature via standard minimally invasive access technique. The SoundBite XS Crossing System facilitates intraluminal placement of conventional guidewires or treatment devices beyond the peripheral arterial CTO.

XS Console
Generates the Shock Waves
- Mobile, non-sterile, reusable
- Intuitive, easy-to-use interface

Active Wire XS 14P
Waveguide that Delivers IVL to Lesion
- Sterile, single-use
- Specifications: 0.014” max outer diameter over usable length, 175cm usable length
- Accessories – disposable accessories to assist in crossing

Torquer
Side-mounted torquing device to provide gripping surface during handling
- Sterile, single-use device
- Used with physician discretion

Curving Tool
Provides Different Curve
Options for Wire Tip
- Sterile, single-use
- Used to create a short or long gradual curve on distal tip of the Active Wire XS 14P
- Used with physician discretion
Breaking New Ground with Active Wire
Promising Early Performance in Highly Calcified Lower Extremity CTOs
We are committed to building our clinical evidence with our devices in support of better utilization and procedure results and outcomes for patients suffering from calcific occlusive cardiovascular disease.
The PROSPECTOR Study1
Prospective, multi-center, single-arm pivotal study in Europe and Canada.
Ongoing Clinical Trials
CaTO PAD – Currently Enrolling
Prospective, multi-center, non-randomized, clinical study evaluating the performance of the SoundBite XS Crossing System-14P in the US and Europe.
Learn MoreTo learn more, review publications.
A Multinational, Single-Arm Pivotal Study Assessing the Performance of the SoundBite Crossing System for Peripheral Chronic Total Occlusions (The PROSPECTOR Study)
Recanalization of CTOs with SoundBite Active Wire
Novel Crossing System for the Recanalization of Complex Chronic Total Occlusions: Ex vivo Proof of Concept of the SoundBite Crossing System.
Novel Crossing System for Chronic Total Occlusion Recanalization: First-in-Man Experience With the SoundBite Crossing System
References:
- Therasse, E. et al. A Multinational, Single-Arm Pivotal Study Assessing the Performance of the SoundBite Crossing System for Peripheral Chronic Total Occlusions (The PROSPECTOR Study), J Vasc Interv Radiol. 2022; 33:50-59.
